However, Integrity Applications is planning to submit the device for trials to gain approved status. 
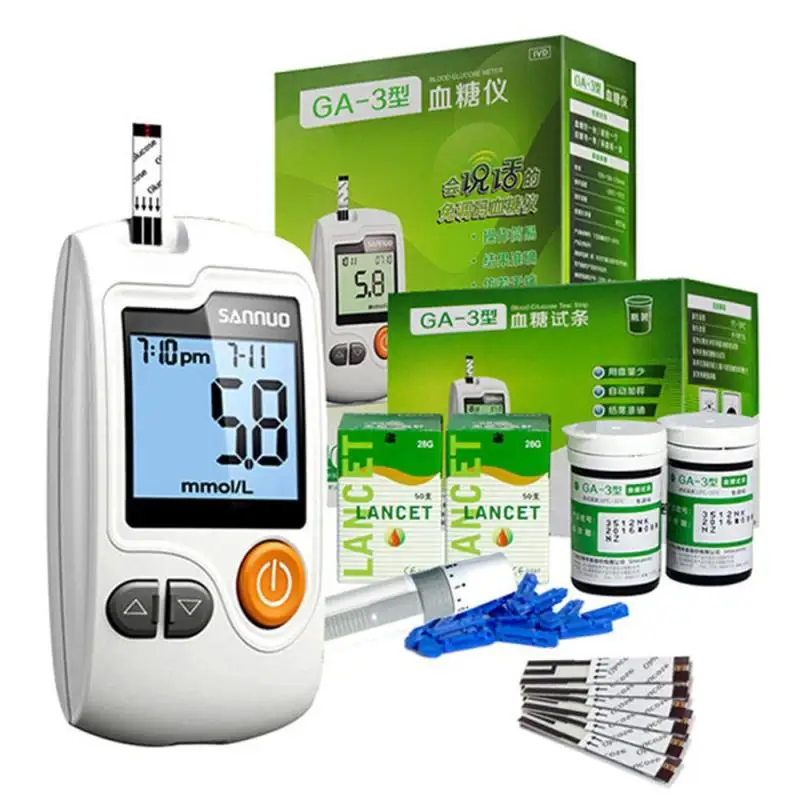
This battery-operated noninvasive device helps people with type 2 diabetes monitor their blood glucose levels through their ear lobes.Īt the time of publication, these tests do not have approval from the Food and Drug Administration (FDA). Integrity Applications, a company that focuses on needle-free blood glucose monitoring, manufactures GlucoTrack. However, according to 2019 research, earlobe scanning is not as effective for people with type 1 diabetes, especially during periods of exercise or very low blood sugar. Some monitors can check blood glucose levels through the ear lobes.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
